BACKGROUND
Chronic kidney disease (CKD) is known to have significant morbimortality worldwide. In 2017, there were 697 million patients with all-stage CKD, for a global prevalence of around 10%, which represents an increase of 30% since 1990, with differences depending on the geographic region.1 The high mortality associated with CKD has been described since 1970 and the growing interest in this theme is due to the increased prevalence of this disease.2
Patients with CKD, and in particular those with end-stage renal disease (ESRD), typically carry a large burden of comorbidities, and the start of hemodialysis (HD) leads to a higher risk of decompensation.3 In fact, annual mortality rates among HD patients are 10 to 30 times higher than in the general population.4 Various studies have demonstrated that incident patients experience a higher mortality rate within the first 3 to 4 months of dialysis.5-9 Predicting early mortality is important to help in the decision of initiating HD versus conservative care. The rising number of elderly patients initiating HD highlights the importance of having tools to identify patients in whom the burdens of dialysis care may outweigh its benefits.10-11 Currently, there are some validated scores to predict mortality, although only a few evaluate early mortality.12-14 Therefore, our objectives were to calculate early mortality rate in a population of incident HD patients, characterize the associated risk factors and develop a predictive model for early mortality.
METHODS
This study followed the STROBE statement for observational studies.15 The Ethics Board of our hospital approved the study prior tocommencement. Requirement for individual informed consent waswaived.
STUDY DESIGN, SETTING AND PARTICIPANTS
We performed a single-center retrospective population-based casecontrol(1:3) study of all incident adult patients at the HD unit of Centro Hospitalar Lisboa Ocidental, between 1st of January 2013 and 31st of December 2018. This tertiary care hospital serves a population of about 450000 inhabitants.16 All incident HD patients from this área start their treatment at our institution’s HD unit, according to local policy, and irrespective of their burden of disease or clinical condition.
Inclusion criteria were: 1) Age ≥18 years; 2) HD start between 1st of January 2013 and 31st of December 2018 at Centro Hospitalar Lisboa Ocidental HD unit. Exclusion criteria were; 1) Age <18 years 2) Switch of modality from HD to peritoneal dialysis (PD) or kidney transplant in the first 90 days of HD treatment.
OPERATIONAL DEFINITIONS
Early mortality was defined as mortality from any cause within the first 90 days of treatment and chosen as the primary outcome. For each case, three controls were randomly selected. Controls were patients with CKD stage 5 on regular HD for more than 90 days. The authors decided to match cases and controls according to year of start of HD to account for differences in medical practice.
DATA COLLECTION
Data were obtained from review of hospital medical records. Chosen variables included sociodemographic data, institutionalization status, dependence in daily living activities, comorbidities (congestive heart failure, ischemic cardiomyopathy, cancer, chronic obstructive pulmonary disease (COPD), peripheral vascular disease, diabetes mellitus, cerebrovascular disease, arterial hypertension, chronic liver disease, depression, dementia). The Charlson comorbidity index (CCI) was calculated based on patients’ comorbidities.17
Decision of renal replacement therapy initiation was up to the discretion of the nephrologist. Variables related to renal care included nephrologist appointment at least 90 days prior to ESRD, CKD etiology, ESRD due to non-recovery from acute kidney injury (AKI) (dialysis dependence beyond 90 days after AKI), emergency initiation of HD (defined as any first treatment started due to an emergency condition or not appropriate to delay for >24h), vascular access (fistula/graft or catheter), and laboratory data at HD initiation [hemoglobina (g/dL), serum albumin (g/dL), serum creatinine (mg/dL), eGFR (mL/min/1.73m2, calculated using the 2009 CKD Epidemiology creatinine equation), iPTH (pg/mL), ferritin (ng/mL), calcium (mg/dL), and phosphorus (mg/dL)].
Patients were followed from enrollment until the first of the following events: death, transplant, switch to PD or end of the study period.
STATISTICAL ANALYSIS
Descriptive statistics were calculated and expressed as mean (± standard deviation [SD]) or median (interquartile range [IQR]) for parametric and non-parametric continuous variables, and count (%) for categorical variables, respectively. We compared variables between survivors and non-survivors at 90 days after initiation of HD by using Student’s t-test, Mann-Whitney U test, or Fisher’s exact test, where appropriate. In the event of missing data values or loss to follow-up, data were not replaced or estimated. Multivariate logistic regression was used to calculate the adjusted odds ratio (aOR) with 95% confidence intervals (CI) for the variables associated with early mortality. We assessed for collinearity. All continuous independente variables were found to be linearly related to the logit of the dependente variable. In addition to variables associated with each outcome on exploratory univariate analysis (with a level of significance p<0.05), those with biological plausibility were incorporated in the multivariate model. We excluded candidate predictors with missing data. To reduce the risk of false positive findings and improve model performance we used the events per variable rule of thumb of 10.18-19 Modelling was performed using a conventional backward elimination based on the likelihood ratio test statistic. Collinearity was avoided where appropriate.
Model performance and calibration were assessed by the Hosmer-Lemeshow goodness-of-fit test and discrimination by the area under the receiver operative characteristic (AUROC) curve. We validated the prediction score internally using the bias-corrected and accelerated (BCa) bootstrap method in the original data set by sampling with replacement for 1000 iterations.20-21 A p<0.05 was considered statistically significant for all comparisons.
Statistical analysis was performed using SPSS version 23.0 (IBM Corp, Armonk NY).
RESULTS
Our study cohort included 626 incident HD patients. Among those, 48 cases of early mortality were identified (7.7%), ranging from 5.5-9.2%, depending on the year. Controls were randomly selected, with a total of 144 controls. (Figure 1)
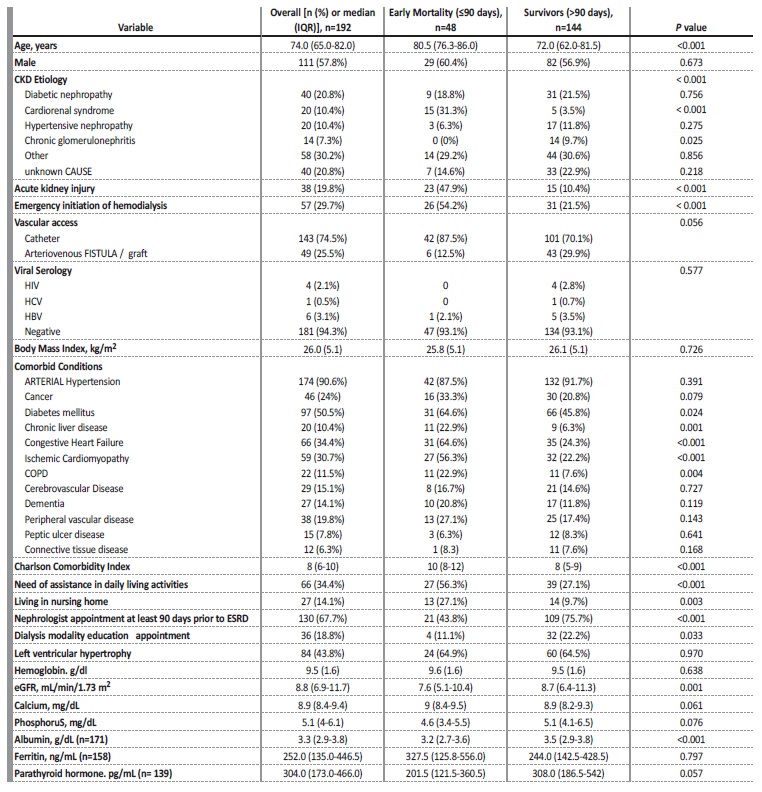
Table I shows baseline characteristics and comparison between survivors and non-survivors at 90 days after initiation of HD. Median (IQR, years) age was 74 (65-82), 57.8% (n=111) were male, the majority (74.5%, n=143) used a central venous catheter as the initial vascular access and the most common etiology of CKD was diabetic nephropathy (20.8%, n=40). 57 patients (30%) had emergency HD initiation and 38 patients (20%) had non-recovering AKI leading to ESRD.
UNIVARIATE LOGISTIC REGRESSION ANALYSIS
Briefly, patients who died were more likely to be older [OR 1.07, 95% CI (1.03-1.11); p<0.001] and have cardiorenal syndrome [OR 12.64, 95% CI (4.29-37.25); p<0.001] or non-recovery from AKI [OR 7.91, 95% CI (3.63-17.24); p<0.001] as cause of ESRD. Patients with early mortality tended to start HD in an unplanned manner [OR 4.31, 95% CI (2.15-8.62); p<0.001], to have need of assistance in daily living activities [OR 3.46, 95% CI (1.76-6.82); p<0.001], to live in nursing homes [OR 3.45, 95% CI (1.49-8.01); p=0.004], to have diabetes mellitus [OR 2.16, 95% CI (1.10-4.24); p=0.026], chronic liver disease [OR 4.46, 95% CI (1.72- 11.57); p=0.002], congestive heart failure [OR 5.68, 95% CI (2.81-11.48); p<0.001], ischemic cardiomyopathy [OR 4.50, 95% CI (2.25-8.99); p<0.001], COPD [OR 12.64, 95% CI (4.29-37.25); p<0.001], and higher CCI score [OR 1.47, 95% CI (1.27-1.70); p<0.001]. Patients were less likely to have 90-day mortality if they had nephrologist appointments at least 90 days prior to ESRD [OR 0.25, 95% CI (0.13-0.50); p<0.001] or a higher serum albumin [OR 0.34, 95% CI (0.19-0.62); p<0.001].
MULTIVARIATE LOGISTIC REGRESSION ANALYSIS
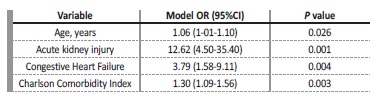
Multivariate logistic regression analysis with backward elimination procedure showed that the following variables were retained in our model (Table II): older age [aOR 1.06 (1-01-1.10), p=0.022], ESRD due to non-recovery from AKI [aOR 12.62 (4.50-35.40), p<0.001], congestive heart failure [aOR 3.79 (1.58-9.11), p=0.003], and CCI score [aOR 1.30 (1.09-1.56), p=0.005]. The model showed an excellent level of discrimination [AUROC 0.881 (95% CI, 0.827 to 0.935).
DISCUSSION
We performed a retrospective cohort study of all incident HD patients from a large Portuguese population to describe the incidence and risk factors associated with early mortality.
KEY FINDINGS
Early mortality occurred in 48 (7.7%) cases of our cohort. We identified older age, ESRD due to non-recovery from AKI, congestive heart failure, and higher CCI score as early mortality predictors.
COMPARISON TO PREVIOUS STUDIES AND INTERPRETATION OF RESULTS
Early mortality varies from 5.5 to 9.4%, depending on the year considered. ERA-EDTA Registry reported an annual mortality rate of 15.9% and early mortality of 5.3%22, lower than USA’s data on annual and 90-day mortality rate: 18.5-19.9% and 6.6-7.2%, respectively.23 Portugal is one of the European countries with higher prevalence of ESRD. During our study period, the national incidence rate of HD was 202-230/pmp and the gross annual and 90-day mortality rates were 12-13.5% and 4-6%, respectively.24 Our study showed an early mortality rate slightly higher than previously reported in national and European registries, but similar to the rate reported in the USA. Patients in our cohort were older, with a mean age of 71.6 vs a mean age of 68 in national and international reports and had a higher percentage of central venous catheters, which is a well identified mortality risk factor (25.5% vs 49%).(23 It has been recognized that elderly patients are more prone to adverse outcomes after HD initiation due to higher percentage of frailty.10,24 More than half the cases of early mortality were in patients with advanced age (>80 years-old) and extensive comorbidities (CCI ≥9), probably due to the presence of frailty syndrome, which is associated with higher mortality.
Our model identified older age, ESRD due to non-recovery from AKI, congestive heart failure, and CCI score as early mortality predictors. Older age and comorbidities, particularly heart failure, have been described in most of the studies published as risk factors associated with early mortality.5-8,11-14,26-28 However, most studies and predictive scores have defined early mortality as death occurring in the first 180 days of HD. We decided to use a 90 day-threshold since there is a known peak in mortality in this interval of time12,14 and because this timing is used in official national and international epidemiological reports.22,23 There are two main studies that investigated early mortality predictors in the first 90 days after HD start, published by Thamer et al.12 and Couchoud et al14.
Couchoud et al.’s study aimed to evaluate early mortality in elderly patients (age over 75 years). Not surprisingly, patients’ median age was higher than in our cohort (81 vs 74), as was the early mortality rate reported (10.5% vs 7.7%). Regarding mortality predictors, male gender, age over 85 years, congestive heart failure, peripheral vascular disease, dysrhythmia, severe behavioral disorders, active malignancy, lower serum albumin, and impaired mobility were independently associated with 90-day mortality. Thamer et al. also studied mortality predictors in older patients (age over 67 years) and developed two predictive models: a comprehensive one and a simple risk score, with 7 variables, based on the first one. The simple model showed that patients who died in the first 90 days were more likely to be older; to need assistance with daily living activities or be institutionalized; to have lower érum albumin; to have cancer, congestive heart failure and to have a higher number of hospitalizations. Our model has CCI score as an independent mortality predictor, which illustrates a high burden of comorbidities. Age and congestive heart failure were also predictors of early mortality in our model, similar to what was seen in both published models. Interestingly, both studies identified impaired mobility or need for assistance in daily living activities as an independent risk factor for early mortality. Functional and cognitive impairment is highly prevalent in patients reaching ESRD and strongly associated with adverse health outcomes, as shown in most studies.8,12-14,29-31 While we only found it to be a risk factor for early mortality in univariate analysis and recognize that the definition used might be different from ours, we highlight the importance of including measures of disability in the evaluation of incident patients in HD. Unlike Thamer et al, we did not include hospitalizations, due to the impossibility of accurately ascertaining the number of hospitalizations by examining the patients’ clinical records.
Heart failure as an independent risk for mortality in HD patients, whether incident or chronic, has been studied and confirmed.32-36 Among HD patients, those who have cardiovascular disease have worse prognosis. Cardiovascular disease is the most common cause of death in HD patients. In our study, we showed that heart failure is associated with a four-fold increase in the risk of early mortality.
Several studies have also shown that patients with heart failure that develop acute or acute-on-chronic kidney disease with HD dependence have a poorer prognosis, with a median survival of less than 4 months.36-37 AKI requiring dialysis is associated with substantial morbidity, mortality, and progression to CKD. Patients are considered to have reached ESRD after 90 days of AKI with dialysis requirement.38 Non-recovering AKI is a known precipitant of ESRD, although studies evaluating its burden on early mortality are lacking. Our report shows that nonrecovering AKI is as an independent predictor of early mortality, and this finding maintains its significance even after adjusting for emergency HD start, suggesting that the acute disruption of hormonal and metabolic homeostasis associated with AKI plays a pivotal role in determining worse outcomes, even when adjusting for the severity of disease.
Although not incorporated in our model, there are some risk factos that were found to be independently associated with early death. Patients with lower serum albumin were found to have a higher probability of dying during the first 90 days of treatment. While most studies consider hypoalbuminemia a modifiable risk factor6,12-14,27-28, we and others consider that the correlation between albumin and mortality is largely dependent on concomitant systemic inflammation and not amenable to intervention.39 Predialysis care, namely nephrology appointments for more than 90 days prior to ESRD, seems to confer some protection in terms of survival odds, due to the possibility of having a well-informed patient starting dialysis in a planned manner, with an arteriovenous fistula as vascular access, which is known to confer a survival advantage in incident HD patients.40,41 Improving predialysis care could lead to a decrease in emergency HD initiation due to lack of follow-up, another significant variable, as shown in multiple studies.42-45
Our model is significant in decision making. Incorporating predictive models into CKD management is useful to inform patients and families about ESRD treatment options and provide a more patientcentered approach. None of the variables in our model are modifiable, so they could be useful to identify those who have an unacceptably high risk of starting renal replacement therapy and would have a higher quality of life on a more conservative approach.25
STUDY LIMITATIONS
Our study has several limitations that warrant consideration. Firstly, since our study is retrospective and single-center, it is potentially predisposed to bias and residual confounding, along with limited generalizability.
Secondly, there were no predefined criteria for renal replacement therapy initiation, which was up to the discretion of the treating nephrologist. Thirdly, our model lacks external validation. Finally, since we did not include a control group of patients a priori excluded for RRT, we cannot definitely ascertain the impact of chronic dialysis on survival among high-risk patients.
CONCLUSIONS
In conclusion, we have developed a predictive risk score for early mortality in patients who initiate HD, which is in agreement with published national and international studies. Incorporating this prediction model into CKD management may help to inform patients about ESRD treatment options and provide a more patient-centered approach to care. In the future, research should be conducted to externally validate our findings, possibly by developing a nationwide registry of patients that were considered ineligible for chronic HD or opted for conservative management and exploring their baseline characteristics and outcomes.46