INTRODUCTION
Brazil has great biodiversity, but many species still require study, such as the fruit species of the Cerrado, including Campomanesia adamantium (Cambess.) O. Berg. (Arantes and Monteiro, 2002), popularly known as gabiroba. Campomanesia adamantium belongs to the Myrtaceae family, and Brazil has 24 genera and 975 species of this fruit (Sobral et al., 2013). This family has been the focus of studies in the Atlantic Forest, the Araucaria forests, and even in the recovery of mining areas (Garcia et al., 2009; Danner et al., 2010; Lourenço and Barbosa, 2012), as well as histochemical studies (Kuster and Vale, 2016).
The gabirobeira is described as a 0.3- to 2-m tall shrub with fruiting between the months of September to November, producing globular fruit 2 to 2.5 cm in diameter, with yellow flesh when ripe (Porto and Gulias, 2010), and has antimicrobialactivity, with high antibacterial and antifungal potential (Sá et al., 2018). It is necessary to study of Cerrado plants, as well as phenolog, due to the loss of natural vegetation, but in recent years more than 50% of its area has been converted to grain crops and pastures (Guilherme et al., 2011; Franco et al., 2014; Rezende-Silva et al., 2019).
Phenology is dedicated to the observation of plant cycles (Staggemeier et al., 2010), the repetition of biological events, and the causes of these cycles in relation to abiotic selective factors, such as temperature, precipitation, and photoperiod, and also in relation to biotic factors, such as seed dispersal syndromes, which help us to understand the spatial dispersion of the plants. Phenology is also dedicated to the understanding of the interrelationship between the phenological phases characterised by these repetitive events within one or multiple species (Lieth, 1974; Cortés-Flores et al., 2013).
Knowledge of the distribution of the types of vegetation and their phenological variations are indispensable in programs aimed at developing and understanding the coexistence of different ecosystems, whether natural, agricultural, or industrial (Canavesi et al., 2005). It is also essential to better understand the ecology and evolution of species and communities (Newstrom et al., 1994).
Phenology is very important also to the botanical and ecological points of view, contributing directly or indirectly to the development and understanding of projects and research in forestry. Phenology is also a fundamental tool in the management and restoration of degraded and riparian areas (Santos and Ferreira, 2013).
Campomanesia adamantium is one species with high economic potential due to the varied potential uses of fruits and other plant parts. However like most native Cerrado species, there is a lack of agronomic information that will enable the start of commercial crops, in this regard the information on growth and development of plants of this species should be prioritized (Araújo and Souza, 2018).
Several researchers have given great importance to external factors, such as seasonality, periodicity and timing, which act as triggers for endogenous factors that trigger the vegetative and reproductive phenological phases (Janzen, 1967).
Another external factor studied in fruit is the photosynthetically active radiation received by olives (Cherbiy-Hoffmann et al., 2013), apples (González-Talice et al., 2013), blueberries (Lobos et al., 2013), strawberries (Costa et al., 2011) and melons (Pereira et al., 2010). The shading of a plant may cause changes in the microclimate in which it is located and, according to the time and intensity, there may be physiological, anatomical, and reproductive changes that negatively affect fruit production (Mitchell, 2003).
The yield of a crop, crop genetics, and other conditions are directly linked to the efficiency of photosynthetic radiation (Morais et al., 2009). Thus, the production of a culture depends on effective interception of the incident solar radiation on the photosynthetic plant organs, its conversion into energy, and translocation of assimilates in the different plant organs (Fereres, 1995).
In view of this, the aim of this study was to evaluate the time of occurrence, intensity of reproductive and vegetative phenological phases, and their correlations with climate variables, such as temperature, humidity, and precipitation, and the influence of photosynthetically active radiation in the fruit Campomanesia adamantium (Cambess.) O. Berg.
MATERIALS AND METHODS
Study site
The phenological study and growth of Campomanesia adamantium was conducted of Rio Verde, Goiás, Brasil, located at the coordinates of latitude 17 ° 56' 46 '' S and longitude 51 ° 11 '50' 'W, at an elevation of 694 m.
The location studied is in the phytogeographical area of Cerrado, or cerrado sensu lato, and the cerradão is the predominant vegetation type on site (Veloso et al., 1991).
According to Köppen, the region falls within the Aw climate type, characteristic of tropical humid climates, with two distinct seasons: dry in winter and humid in the summer. The average annual rainfall is between 1200 and 1800 mm. The rainy season extends from November to March, with the wettest quarter corresponding to the months of January, February, and March. The dry period is represented by the months of June, July, and August, with May and September as the months of transition between the dry and wet seasons, respectively. The average maximum temperatures are around 18 °C, and the annual temperature range is less than 4 °C (SEPLAN-GO, 2005; EPE, 2006).
Phenological data collection
The phenological characterisation was made in ten Campomanesia adamantium adults, which were numbered, labelled, and evaluated in terms of age. The inclusion criteria for the plants were that the diameter ar breast height (30 cm ground) was ≥ 3 cm and the plants were in the reproductive stage.
The collected botanical material was deposited in the Green River Herbarium (IFRV) of the Federal Institute Goiano - Campus Rio Verde. The species identification was based on the scientific literature and consultation with experts.
Phenological observations of vegetative phenological phases (shoot, young leaf, adult leaf, and senescent leaf) were made monthly and reproductive phenological phases (button, anthesis, immature fruit, and ripe fruit) weekly. The observations of vegetative and reproductive phenological phases occurred between October 2012 and September 2013, always by the same observer.
The phenological events were recorded by the intensity ratios and activity phenological phases. The intensity index of phenophase in each individual was verified by the Fournier method using a semiquantitative scale interval of five categories (0-4), with an interval of 25% in each category, as follows: 0 = absence of phenology; 1 = presence of phenophase of magnitude of 1-25%; 2 = presence of phenophase of magnitude 26-50%; 3 = presence of phenophase with magnitude 51 to 75%; and 4 = presence of phenophase of magnitude 76-100%. The activity index revealed the presence or absence of phenophase in each individual (Bencke and Morellato, 2002).
To calculate the intensity index, the intensity values for all individuals of each species were summed and divided by the maximum possible value (number of individuals multiplied by four) each month. The obtained value was then multiplied by 100 to turn it into a percentage value (Fournier, 1974; Bencke and Morellato, 2002). The activity index method indicates the percentage of individuals in the population that is manifesting certain phenological events. This method can also be used to calculate the timing of individuals in a population (Morellato et al., 1990), taking into account that the greater the number of individuals simultaneously expressing phenophase, the greater the timing of this population.
The environmental variables for the study period were obtained from the Meteorological UniRV- University of Rio Verde Station and correlated to the studied phenological phases based on the Spearman correlation coefficient (rs).
Biometric data collection
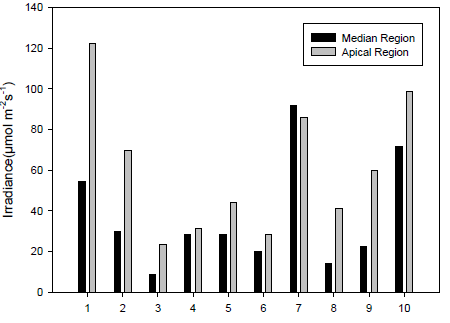
The photosynthetically active radiation (PAR) levied on each individual was measured weekly by radiation sensors (Decagon Devices, Pullan, USA) in the middle region of the crown and apex of the region (Figure 1). From these data, two branches were marked in each individual: a branch with a higher incidence of light on the plant apex and a branch with a lower incidence in the middle region of the plant, called the sun and shade branches, respectively.
Prior to biometric evaluation monitoring, the total number of fruits per branch (sun and shade) were counted. Five fruits were selected at random in the middle portion of each branch for further monitoring of the longitudinal diameter and transverse diameter of the fruit to full maturity. Measurements were made with a hand calliper with an accuracy of 0.02 mm.
The actual fruit set was checked weekly by the total number of fruits counted in the sun and shade branches.
For fruit biometrics, the experiment was conducted in an experimental design that consisted of randomised blocks and the means and the standard error of the mean were obtained. The correlation between the number of fruits and the diameters was tested by Pearson correlation, using Bioestat software.
RESULTS
Vegetative Phenological phases
Shoots were observed in almost all of the evaluation period, except for the months of March, May, and August. The largest shoot activity peaks were observed in October, November, and January 2012 and September 2013 (Figure 2), and the highest percentage of individuals presenting shoots occurred in September, along with most of the canopy coverage ratio (Figure 2B).
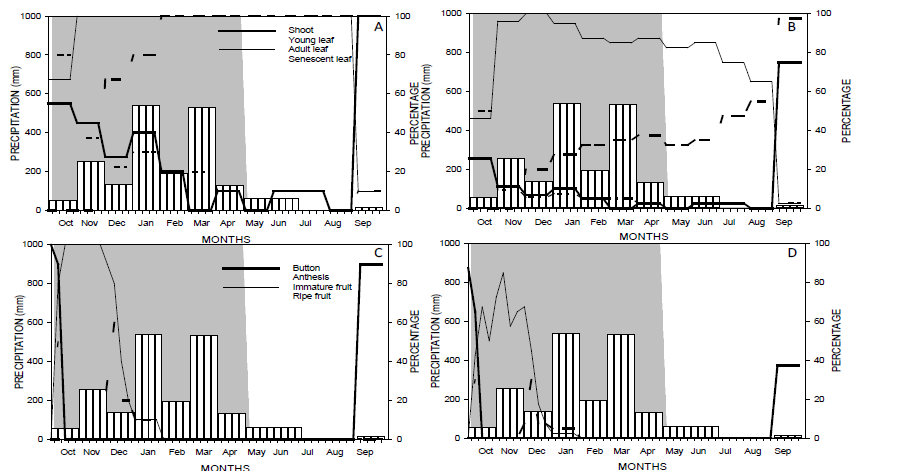
Figure 2 Distribution of precipitation and phenophases studied in Campomanesia adamantium (Cambess.) O. Berg. According to the activity index of the vegetative (A) and reproductive (C) phenological phases and with the intensity index of vegetative (B ) and reproductive (D) phenophases. Shoot and button phenophases (continuous thick line); young leaf and anthesis (lower dashed line); adult leaf and immature fruit (continuous thin line); and senescent leaf and ripe fruit (larger dashed line). The gray area corresponds to the wet season.
The young leaf activity was recorded between the months of October and April and the months of June, July, and September; this activity decreased as time went on (Figure 2A). The maximum activity was recorded in October, together with greater intensity; however, after this period, intensity values declined (Figure 2B).
The adult leaves were less active during the first three weeks of evaluation and only after the month of November did the adult leaves reach maximum activity; the period of maximum activity extended until the month of August and decreased thereafter (Figure 2A). The intensity of mature leaves was inversely proportional to the intensity of young leaves. For mature leaf intensity, lower values were observed in October, followed by an increase in the percentage with maximum values in December, with a slow decrease afterward (Figure 2B).
The senescent leaf was observed from December 2012 and the maximum values of fall activity occurred between February and September (Figure 2A). The leaves’ intensity decrease was zero in October and November 2012; intensity increases from December 2012 to almost 100% in September 2013 (Figure 2B).
Reproductive Phenological phases
The button activity, Figure 2C, and the intensity, Figure 2D, were only observed in October 2012 and September 2013 (Figure 2C). At the beginning of October these features were more prominent in relation to the end of the month, making it outstanding at the beginning of September.
Anthesis activity was observed only in October 2012 and the peak of this activity was reached in the second week of the month (Figure 2C). The intensity of anthesis did not cover more than 40% of the cup (Figure 2D).
Fruiting occurred from the third week of October 2012 to January 2013, evidenced by the presence of immature fruit. The maximum activity of immature fruits was observed between the third and the last weeks of October and November, respectively (Figure 2C). The higher intensities of immature fruits were observed in the months of October, November, and December, with values of 67.5%, 72.5%, and 67.5%, respectively (Figure 2D). The intensity of 2.5% immature fruit was checked in January 2013 (Figure 2D).
The activity of ripe fruits was observed only in the months of December and January, with a peak in December (Figure 2C). The intensity of ripe fruit in the month of December was approximately 40%, which is the maximum value reached during the evaluation period (Figure 2D).
Correlation between phenology and climatic factors
During the study period, the average temperature in the months ranged from 21.3 °C to 32.7 °C. The highest average temperatures, recorded in July, August, and September, were 32, 26, and 25 °C, respectively. The relative humidity ranged between 41.5% and 83.6%. The highest relative humidity was found in January (83.6%), followed by February (76.9%) and March (75.7%). Precipitation changed by 0.0 mm to 538.8 mm over the study period. The highest rainfall was recorded in January, followed by March, whereas precipitation was zero in August.
Table 1 shows the correlation values of phenology (activity and intensity of vegetative and reproductive phenological phases) with the climatic factors (temperature, humidity, and precipitation).
Table 1 Values of Spearman correlations between vegetative and reproductive phenophases with the weather conditions, temperature, humidity, and precipitation
| Phenophases | Temperature | Humidity | Precipitation | |||||||||||||||||||||||||||||
| Activity | Shoot | 0.4978** | -0.4868** | -0.3454* | Anthesis | 0.1489 | -0.2975* | -0.1872 | Immature F. | -0.0209 | 0.2582 | 0.3948** | Ripe F. | -0.2161 | 0.4828** | 0.4305** | Button | 0.2217 | 0.0073 | 0.1244 | Young Leaf | -0.0052 | 0.4356** | 0.5973** | Adult Leaf | -0.5061** | 0.4852** | 0.3484* | Senescent Leaf | -0.0194 | -0.1598 | -0.3468* |
| Intensity | Shoot | 0.4930** | -0.4854** | -0.3436* | Anthesis | 0.1489 | -0.2975* | -0.1872 | Immature F. | -0.0291 | 0.2585 | 0.4027** | Ripe F. | -0.2155 | 0.4826** | 0.4306** | Button | 0.2237 | 0.0032 | 0.1217 | Young Leaf | -0.0036 | 0.4337** | 0.5973** | Adult Leaf | -0.5019** | 0.7822** | 0.7645** | Senescent Leaf | 0.3862** | -0.4550** | -0.5738** |
A significant variation between the correlated factors is indicated (* p = 0.05 and ** p≤0.01).
The activity and the button of intensity showed significant correlations with temperature, relative humidity, and precipitation: positive when correlated with temperature and negative for relative humidity and precipitation.
The activity and intensity of anthesis were negatively correlated with the relative humidity.
The activity and intensity of immature fruits was significantly positively correlated with precipitation.
Significant positive correlations were observed for both the activity and intensity of ripe fruit with humidity and precipitation.
The activity and intensity of shoots were not correlated with any of the variables.
Young leaf activity and intensity were significantly positively correlated with relative humidity and precipitation.
The activity and intensity of adult leaves were significantly positively correlated with moisture and precipitation, but negatively correlated to activity in relation to temperature.
A significant negative correlation was found between senescence and precipitation, as well as the fall in intensity with relative humidity and precipitation; the decrease in intensity correlated positively with temperature.
Biometry of fruits
A higher number of fruits were observed in sun branches than in shadow branches. On average, in the first assessment, each sun branch had 55 fruits, while the shadow branches had 28 fruits on average (Figure 3A).
In both branches, sun and shade, the number of fruits decreased as they developed.
The longitudinal diameter, Figure 3B, and the transverse diameter of the fruit, Figure 3C, increased with increased development.
No differences were observed in the longitudinal diameter values between fruit from the sun and shade branches up to the 7th day; afterward, fruit from sun branches showed higher values.
For the transverse diameter, differences were also identified only on days 7, 14, 35, and 49 after fruiting; the other days, the sun fruits showed higher values. On the last day of evaluation, we observed a reduction in the diameter of the fruits grown under shade.
The initial longitudinal diameter of the sun fruits was 4.1 mm and that of the shadow fruits was 3.5 mm; the maximum values were 14.5 and 13.0 mm, respectively. The initial transverse diameters were 3.7 mm and 3.0 mm for the fruits of the sun and the shadow, respectively; their maximums were 13.6 and 12.4 mm, respectively.
Significant negative correlations were found between the number of sun and shade fruits and their longitudinal and transverse diameters (Table 2).
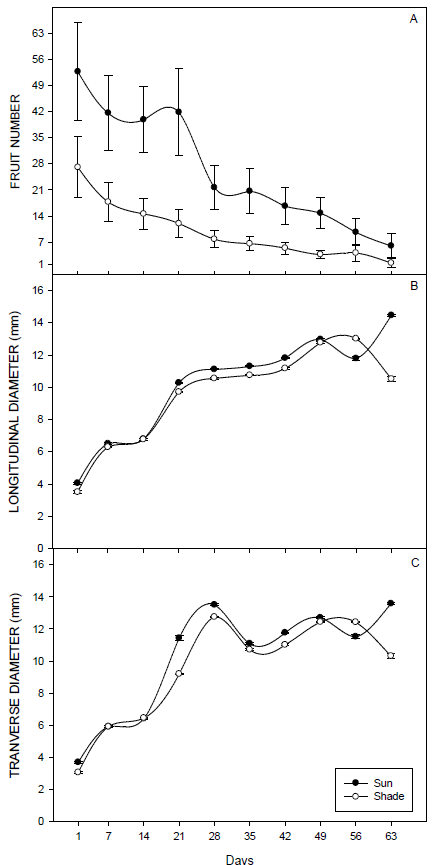
Figure 3 Values corresponding to the number (A), longitudinal diameter (mm) (B), and transverse diameter (mm) (C) of fruits developed in branches of sun and shade. Rio Verde, GO, 2012/2013.
Table 2 Values of the Pearson correlations between the number of fruits of sun and shade and longitudinal and transverse diameters
| D. Longitudinal | D. Transverse | |
| Number of sun fruits | -0.9196** | -0.8403* |
| Number of shade fruit | -0.9410** | -0.9188** |
A significant variation between the correlated factors is indicated (* p = 0.05 and ** p≤0.001)
DISCUSSION
The peak occurrence or shoot activity, along with higher intensity during the month of September, took place during a period of lower rainfall. Similar results were found by Braga et al. (2010), who studied four native Cerrado species, two of which belonged to the Myrtaceae family, which presented leaf drop and sprouting during the dry season.
The lack of a significant correlation of shoot activity and intensity with the studied variables indicates that the temperature, relative humidity, and precipitation are not the main factors that boost budding. Different results were found by Amorim et al. (2010), who found higher shoot values in months of higher temperatures in species belonging to the Myrtaceae family.
The non-occurrence of a positive correlation between shoots and precipitation is probably be related to the ability of Cerrado plants to develop deep roots with exclusive access to the denser layers of the soil and thus have moisture throughout the year; this would be a way for species present in seasonal environments to survive (Canadell et al., 1996).
The plants have organs called sources and drains. The source body, from photosynthesis, produces assimilates that can be used as a source of energy in breathing or be transported and stored in organs reserves, better known as drains. The main source organs are the adult leaves and the drains are the young leaves, roots, meristems and fruits, which, with development, became a source organ (Duarte and Peil, 2010). The shoots’ and young leaves’ activity decreased across the study period, indicating development into adult leaves. The leaves begin to develop as drain organs, assimilate importers, and transitions into a drain source; increasing shoot sprouts in September indicates a new cycle of the plant.
The activity and the intensity of young leaves was inversely proportional to the activity and the intensity of mature leaves due to leaf development, which, in time, moved from mature leaves to young leaves. The activity and the intensity of young leaves had a significant positive correlation with moisture and precipitation, indicating that increased precipitation increased the number of young leaves. Correlations between precipitation and young leaves have been found in other vegetative species, such as Anadenantehra macrocarpa (Benth.) Brenan (Silva et al., 2012).
A significant positive correlation of adult leaves with moisture and precipitation and a negative correlation for the temperature indicates that with increased humidity and precipitation and reduced temperature, there was a higher incidence of adult leaves. A high humidity environment favours leaf expansion and stomatal aperture, allowing greater CO2 assimilation with consequent production of assimilates, thus aiding in the transition of young leaves to mature leaves.
The senescent leaf activity was negatively correlated with precipitation, revealing that decreasing precipitation increased leaf drop. The decline in intensity was positively correlated with temperature and negatively correlated with humidity and rainfall, indicating that more falling leaves were found with higher temperatures and lower humidity and rainfall. The negative correlation between precipitation and leaf fall was also found in other species, as reported by Silva et al. (2012).
Leaf abscission is directly related to the production of ethylene, which is the primary signal that drives this process, while auxin reduces the sensitivity of cells to ethylene action (Taiz and Zeiger, 2013). Thus, probably as the temperature increased, the plant suffered from heat stress, triggering an autocatalytic formation of ethylene, leading to leaf abscission. However, the gradual leaf fall during the year may also be the result of a natural senescence or a species adaptation to prevent the loss of water, acting as a defence mechanism (Lenza and Klink, 2006).
In the growing season, the plant initially mobilises stored reserves in its cotyledons or endosperm and builds its rudimentary form by the activity of the apical meristems of the root and the stem, followed by the formation of floral organs (sepals, petals, stamens, and carpel). After this period, there will be a transition to the reproductive period, with consequent formation of the buttons, which will result in flowering (Taiz and Zeiger, 2013).
It was fast and intense button activity for Campomanesia adamantium with increased temperatures and reduced humidity and rainfall, a fact confirmed by Vogado et al. (2009) in Myrtaceae species. The anthesis period was also fast, only in October, though not as intense as the button period. Both the activity and the intensity of anthesis increased with decreasing moisture.
The presence of flowers was observed for the months from August to November in Campomanesia pubescens Martius in cerrado fragments located in the Ecological Park Falls Rio Bonito, in Minas Gerais (Borém, 2009), differing from the data obtained in this study, in which flowering occurred only in October 2012. It has also been reported that Campomanesia adamantium (Cambess.) O. Berg present in the south of Mato Grosso do Sul, shows a peak of flowering from September to October (Nucci and Alves-Júnior, 2017). The identification of flowering periods is important in breeding programs because it allows planning to collect pollen and direct crosses for the species (Danner et al., 2010).
The reproduction of a plant is dependent on the time of occurrence of the phenology of anthesis, because any variation that occurs in this phase will influence the successful pollination and seed production rate (Stephenson, 1981). Some authors point out that anthesis would be triggered by increasing the photoperiod, temperature, and humidity in the transition from the dry season to the wet; flowering in the hottest and rainy season is advantageous due to increased availability of light, nutrients, and pollinating insect activity (Morellato et al., 1989; Morellato, 1991; Morellato and Leitão Filho, 1992).
Although they belong to the same genus, some species may have different behaviour in relation to flowering. In a study on the reproductive phenology of the species Campomanesia xanthocarpa O. Berg and Campomanesia guaviroba (DC.) Kiaersk, the researchers detected the presence of buds or flowers during the period of a year of study for C. xanthocarpa. As for C. guaviroba, the presence of flowers was observed only in February (Staggemeier et al., 2007), revealing behaviours in both species that are different from those of C. adamantium in this study, and although they belong to the same genus, these three species have different behaviours.
In the period of flowering and fruiting, humidity and temperature are the key factors for fruit set. High humidity can adversely affect the retention of the pollen grain by reducing the viscosity of the stigmatic fluid, while the high temperature dries the stigma and, as a result, there is premature fall of flowers and fruits fruit set in phase.
The fruiting of C. adamantium at the beginning of the wet weather event was also observed in Myrcia guianensis, which also belongs to the family Myrtaceae (Vogado et al., 2009). However, the peak of immature and mature fruits occurred in November, the beginning of the rainy season, in Myrcia guianensis (Vogado et al. 2009) and C. adamantium in this study. Only the unripe fruit peaked in November, with the peak of the ripe fruits in December, also in the rainy season.
Divergent results of the fruiting period were found in a study with C. pubescens in Cerrado fragments in Minas Gerais, whose fruiting began in September (Borém, 2009). In species of the same genus but different domains, fruiting occurred in May in C. xanthocarpa and C. guaviroba present in the Atlantic Forest (Staggemeier et al., 2007).
However, in general, the pattern of fruiting of C. adamantium was similar to that of the species of the cerrado fleshy fruits studied by Camargo (2008), who also studied these fruits in the rainy season, as the weather conditions are favourable for fruit development (Batalha and Martins, 2004). According to Santos et al. (2015) the fruiting period of Campomanesia adamantium (Cambess.) O. Berg lasts 63 days, between the months of another and December.
The months with the presence of immature fruits corresponded to the period when the adult leaf activity was higher; thus the adult leaves acted as source organs of assimilates to the fruits and young leaves. The activity and intensity of immature fruits were triggered by increased precipitation, while the ripening of fruits was triggered by the increases in humidity with precipitation. This was unlike Myrcia guianensis, also belonging to the Myrtaceae family, which increases production of immature fruit with increasing temperature (Vogado et al., 2009).
Fruit maturation of C. adamantium was a little later than that of C. xantocarpa, studied in Araucaria Forest, whose fruit ripened at the end of November to the end of December (Danner et al., 2010), while in the present study, there was ripened fruit from December to January for C. adamantium. Knowledge of the fruit maturation period is of great importance because it allows the timing of the harvest in commercial fruit planting (Danner et al., 2010).
Most fruit production in the branches with the highest radiation is according to published studies, which found that artificial lighting promoted an increase in the number of fruits and fruit area of yellow passion fruit (Cavichioli et al., 2006) and Royal Gala apple, and it has been observed that the shading promoted decreases in fruit set and yield (Amarante et al., 2007).
The low yield of fruit can be a consequence of the reduction in the light supply, which reduces the accumulation of reserves and may compromise the differentiation of reproductive buds and fruit set. However, responses to shading can vary by species. This fact was proven in a study with tomato in the north of Minas Gerais, where shaded environments conditioned better responses to the cultivation of the species and did not influence the quality of tomato fruits (Otoni et al., 2012).
The longitudinal and transversal diameter values of shaded fruits are not within the range of values found for C. adamantium (Oliveira et al., 2008). Fruits with lower incidence of radiation had lower biometric values than the fruits with the highest incidence, showing that even within values reported in the literature, fruit on shadowed branches were affected by the low radiation.
Shading also decreased the growth rate of apples, which was attributed by the authors to the simultaneous reduction in the daily amount of sap received by the fruit in shade conditions (Morandi et al., 2011).
When the penetration of solar radiation inside the canopy is affected, there is a reduction of source power and, consequently, the availability of assimilates decreases, thereby reducing the amount intended for fruit and increasing the rate of abortions. This device is for the benefit of the vegetative organs, as there is a stimulating leaf growth to improve the capture of light by canopy (Duarte and Peil, 2010).
The negative correlation between the number of fruits with longitudinal and transverse diameters in both the sun and shade fruits indicates that, insofar as reducing the number of fruit per branch, the fruits present in the branch had increased their longitudinal and transversal diameters. Such a correlation may be connected directly with the distribution of photoassimilates by the relative source and drain, since the photoassimilates are produced to be used as an energy source or as reserves for drain organs, such as fruits (Duarte and Peil, 2010). Thus, to the extent that the number of drain organs decreased, assimilate accumulation was greater, increasing the diameters of the fruits.
CONCLUSION
In the region of Rio Verde, there was young and adult leaf activity in Campomanesia adamantium throughout the study period, except young leaves were inactive in May and August. The presence of sprouts was also quite significant over the period. There was a phenophase cycle of rapid and intense play between October and January for C. adamantium. Shading affected the number of fruits per plant and the longitudinal and transverse diameters of the fruits.