Serviços Personalizados
Journal
Artigo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Acessos
Acessos
Links relacionados
-
 Similares em
SciELO
Similares em
SciELO
Compartilhar
Revista de Ciências Agrárias
versão impressa ISSN 0871-018X
Rev. de Ciências Agrárias vol.42 no.2 Lisboa jun. 2019
https://doi.org/10.19084/rca.17268
ARTIGO
Evaluation of wheat events transformed with the p5cs gene under conditions of water stress
Avaliação de eventos de trigo transformados com o gene p5cs e submetidos à seca
Lettice Aparecida Dias Canette de Lima1, Ivan Schuster2, Antonio Carlos Torres da Costa1 and Eliane C.G. Vendruscolo3,*
1Universidade Estadual do Oeste do Paraná, Departamento de Agronomia, Rua Pernambuco, 1777, CP 91, CEP 85960-000, Marechal Cândido Rondon-PR, Brasil
2Coodetec Cooperativa de Pesquisa Agrícola do Paraná, BR 467,Km 98, CEP 85813-450,Cascavel-PR, Brasil
3Universidade Federal do Paraná, Setor Palotina, Labiogen, Rua Pioneiro, 2153, Jd Dallas, CEP 85950-000,Palotina-PR, Brasil
(*E-mail: egvendru@gmail.com)
ABSTRACT
The enzyme ∆1-pyrroline-5-carboxylate synthetase (P5CS), encoded by the p5cs gene, is the rate-limiting step for proline biosynthesis, which acts as an osmoprotectant. The launch of transgenic commercial varieties depends on physiological and phenotypic characterization of events. The objective of this study was to evaluate five p5cs gene events (591, 14, 326, 164, 312), through physiological analyses and production rates during droughts. Plants of events and irrigated (IC) and stressed controls (SC) in flowering stage were submitted to 16 days of complete water restriction. The following elements were evaluated: relative water content (RWC); malondialdehyde concentration (MDA%); membrane stability index (MSI%); proline content; and production rates. As a result, the effect of water stress was only perceived after 12 days of water restriction with a decrease in the RWC values. Transgenic plants showed higher proline levels on the 16th day, compared to the SC (33 to 62%). The levels of MDA were lower on the 12th and 16th days for all the plants evaluated, but the MSI were similar to the IC for the events 591, 326, and 164, demonstrating the osmoprotectant effect. However, higher levels of proline did not result in higher production rates in the transgenic plants.
Keywords: Poeacea, Abiotic stresses, Proline, Physiological Index, Production
RESUMO
A enzima ∆1-pirrolina-5-carboxilato sintetase (P5CS), codificada pelo gene p5cs, é o passo limitante da biossíntese de prolina, que age como um osmoprotetor. O lançamento de variedades comerciais transgênicas depende da caracterização fisiológica e fenotípica de eventos. O objetivo deste estudo foi avaliar cinco eventos (591, 14, 326, 164, 312) contendo o gene p5cs, através de análises fisiológicas e taxas de produção sob estresse hídrico. Plantas de eventos e controles irrigados e estressados (CI e CS) em estádio de florescimento foram submetidos a 16 dias de restrição hídrica. Foram avaliados: teor relativo de água (RWC); concentração de malondialdeído (MDA%); índice de estabilidade da membrana (MSI%); conteúdo de prolina; e taxas de produção. Como resultado, o efeito do estresse hídrico só foi percebido após 12 dias de restrição hídrica com uma diminuição nos valores de RWC. Plantas transgênicas apresentaram maiores níveis de prolina no 16º dia, comparado ao SC (33 a 62%). Os teores de MDA foram menores no 12º e 16º dias para todas as plantas avaliadas, mas os MSI foram semelhantes ao IC para os eventos 591, 326 e 164, demonstrando o efeito osmoprotetor. Porém, níveis altos de prolina não resultaram em maiores taxas de produção de grãos.
Palavras-chave: Poeacea, estresse abiótico, prolina, índice fisiológico, produção
INTRODUCTION
Wheat is cultivated in approximately 17% of the world’s agricultural land, which corresponds to 200 million hectares, and is one of the major sources of calories and protein in the human diet (Jones, 2005). Due to its significant economic importance, wheat is produced in various countries and climates. Drought and salinity are thought to be the most important abiotic stresses that damage this crop worldwide (Dolferus et al., 2011; Saad et al., 2013).
Water stress promotes loss of cellular water or osmotic stress, and the tolerance capacity is related to the maintenance of cellular integrity, targeting a functioning metabolism, which favors the plants’ recovery after the end of the water deficit period (Ozden et al., 2009).
One of the most important biochemical modifications in plants subjected to abiotic stresses is the production of reactive oxygen species (ROS) (Ahmad et al., 2008). Under water restriction, there is a reduction in the rate of photosynthesis due to the closing of stomata and, along with the water shortage and constant capture of light, the photosynthetic fixation of CO2 is limited – resulting in an accumulation of electrons (Ashraf et al., 2008; Munns & Tester, 2008). The ROS thus produced damage cell membrane through the peroxidation and re-esterification of fatty acids.
Recent studies have shown that genetic manipulation of genes responsible for the biosynthesis of low-molecular-weight metabolites has granted the plants a better tolerance to drought and salinity (Dolferus et al.,2 011; Dunwell, 2014). Proline is an osmoprotectant with a fundamental role in the protection against oxidative damage, due to a more efficient antioxidant system, in which the proline seems to be more involved in the sequestration and inactivation of free radicals and in the protection of antioxidant enzymes (Ozden et al., 2009; Bhaskara et al., 2015). Specifically, the enzyme ∆1-pyrroline-5-carboxylate synthetase (P5CS), encoded by the p5cs gene, is considered to be the rate-limiting step in the synthesis route of proline.
The comparatively high proline accumulation in transgenic plants is responsible to a better growth, more chlorophyll and relative water content and lower levels of lipid peroxidation of plants under osmotic stress (Khan et al., 2015).
Increasing tolerance to water deficit stress, by p5cs genetic transformation, has been obtained for wheat (Vendruscolo et al., 2007; Pavei et al., 2016). Generally, after the transformation, the positive events for the transgenesis are selected according to the presence of a single copy, fertility, and normal phenotypic appearance (Xiao et al., 2009). Several genetic factors can affect transgene expression, including epigenetic factors (Dietz-Pfeilstetter, 2010). The stable expression throughout generations and the non-occurrence of gene silencing seem to be the major challenges for plant improvement in polyploid genomes (Anand et al., 2003; Rooke et al., 2003; Meng et al., 2006; Yao et al., 2006).
In addition, the pre-selection of high productivity events is fundamental for the launch of commercial transgenic varieties, especially when the system of genetic transformation used is the gene gun, which consists of the insertion of gene fragments without the number and position control of insertion in the genome (Mlynorowa et al., 1996; Ulker et al., 1999; Basri, 2005).
This study was carried out with the objective of evaluating the behavior of five transgenic events of wheat containing the exogenous p5cs gene in the 3rd generation (T3), under severe water stress, in order to establish a physiological characterization and an evaluation of the efficiency of transformation events in terms of wheat plants’ tolerance to water stress.
MATERIAL AND METHODS
The experiment for the phenotypic evaluation was conducted in a greenhouse, at Coodetec’s Biotechnology Laboratory, in Cascavel-PR, Brazil. The transformation procedure used in this experiment in wheat (Triticum aestivum L. cv CD200126) was the gene gun. It was executed according to the protocol proposed by Bohorova et al. (1999). The pJS107 plasmid, which contains the cDNA of the p5cs gene (∆1-pyrroline-5-carboxylate synthetase) originated from Vigna aconitifolia (Jacq.) Maréchal was used, driven by the stress-inducible promoter AIPC (Zhu et al., 1998). The pJS107 also contains the gene bar merged into the 35S promoter, which was used for the selection of transformants in the presence of phosphinothricin (PPT) – confirmed by analyses made by Southern Blot (Vendruscolo et al., 2007).
Wheat seeds derived from five transformation events T1 from 12 originally obtained (events 1(164); 2(591); 3(326); 4(312) and 6(14) (Vendruscolo et al., 2007-Figure 1)) were planted in greenhouses under controlled conditions of temperature and irrigation (average temperature 24 ± 2 oC, Relative Humidity 55-75%, watering every day). Seeds of the same genotype (CD200126) were used as a control group, although they were not transformed. The transgenic plants were multiplied and cultivated by self-fertilization until they originated the T3 descent. They were considered homozygous lines for the transgenic insertion through the obtained inheritance pattern of 3:1 in T1 (Chi-squared 1.1 to 3.2).
T3 seeds were placed to germinate in Germitest Paper. After germinating for 3 days, seedlings were transferred into pots (four seedlings per pot), each containing 4kg of previously sifted and fertilized soil (pH 6.4; P = 60 mg.dm-3; K = 1.14 cmol.dm-3; Ca = 6.69 cmol.dm-3; Mg = 3.03 cmol.dm-3; H+Al = 3.18 cmol.dm-3; Cu = 14.45 mg.dm-3; Mn = 400 mg.dm-3; Fe = 21mg.dm-3; Zn = 32.,49 mg.dm-3; SB = 10.86 cmol.dm-3; 41.6 g.dm-3 organic matter). Thinning was applied after 15 days of sowing, remaining only one plant per pot. The experiment involved five transformation events (591, 14, 326, 164, and 312) and the control groups (with and without water stress), in completely randomized design with five repetitions, totaling 35 pots.
When the plants reached the booting stage, around 60 days after germination, corresponding to the stage 45 in the Zadocks scale (Zadocks et al., 1974), water stress was applied and measured by Relative Water Content (RWC%) (Schonfeld et al., 1988). The treatments underwent 16 days of complete water restriction (except for the control group without water stress). For the evaluation of the physiological parameters, the second leaves of all the plants were collected and, for the quantification of proline and malondialdehyde (MDA), flag leaves were sampled every 4 days. After application of water shortage, the pots were irrigated again. The last collection was made four days after the irrigation was restored, totaling 6 collections. The collections of leaves for physiological analyses were always performed in the morning, between 8:30 a.m. and 10 a.m., at an average temperature of 24ºC.
The following parameters were evaluated: proline content (Bates et al., 1973); Membrane Stability Index (MSI) (Babu et al., 2004), and lipids peroxidation level was determined in terms of MDA content (Sairam et al., 1998).
In order to evaluate the production capacity of both the plants of events subjected to water stress and the control group plants, the wheat ears were collected and counted, and the total number of seeds, the weight of 100 seeds, and the total weight of seeds per plant were determined at the end of the cycle.
The results were submitted to analysis of variance (ANOVA) and to the Turkey test to 5% of probability, with the aid of the GENES program (Cruz, 2006).
RESULTS AND DISCUSSION
Although the events used in this study show the insertion of the transgene – confirmed by the Southern Blot in the T1 generation, the number of inserted copies of the gene bar and p5cs has not been determined yet. However, some authors recommend the verification of performance of transgenic events containing multiple copies to confirm the action and effects of the transgene position in drought conditions (Xiao et al., 2009). Yao et al. (2006), for example, reported that the occurrence of transformants with a small number of copies in wheat is relatively rare, especially when the transformation technology used is the gene gun.
In normal water supply conditions before water stress was applied, the relative water contents (RWC) in the control group plants were not different from those in the transgenic plants in the different events evaluated (Figure 1A). Under water stress conditions, the values of RWC were very similar until the 8th day of stress application. A different behavior among the transgenic plants and the control group plants was observed from the 12th day of water stress on. The highest numbers of RWC were found in the events 14, 326, and 164, in the order of 29%, 40%, and 29%, respectively, when compared to the plants under stress of the control group. The RWC numbers decreased even more as the stress was prolonged (16th day), and the events 591, 14, and 326 continued to present a RWC higher than the control group under water stress. The decrease in the RWC demonstrates that the plants experienced water stress as of the 12th day of water restriction. After the end of the water restriction period and plant rehydration (20th day), the plants recovered the RWC numbers to 70% and 80%, including the stressed control group.
Regarding proline concentration, we noted a natural fluctuation in the irrigated control values, which varied from 1.5 to 7 µmols .g-1 of dry weight over the different periods we assessed (Figure 1B). Control and transgenic plants presented proline contents varying from 2.9 to 25 µmols.g-1 of dry weight under water stress in the different events we evaluated. It proves the importance of this osmoprotectant during stress. The events 591 (8th day) and 312 (12th day) presented the highest increase in proline indexes compared to the irrigated controls (2.2 and 4.3 fold, respectively). In control plants submitted to stressed, that increase was 3.7 fold higher on the 12th day compared to the irrigation control. However, we did not register events displaying higher proline contents that could indicate an over-expression due to the presence of p5cs multi copies. In this regard, Rooke et al. (2003) observed that there is no direct correlation between the number of transgene insertions and the expression level.
We observed a decrease in proline levels on the 16th day of water restriction. However, when compared to the control group submitted to water stress, transgenic plants in the different events we evaluated maintained high levels of proline due to the induced-stress expression, as the transgene has the aipc promoter (Vendruscolo et al., 2007). In the assessment administered on the 16th day, the percentages of proline in the transgenic plants in comparison to the control group (under stress) were as follows: 52% (event 591); 63% (event 14); 49% (event 326); 61% (event 164); and 33% (event 312).
The results of proline levels that were different from those of the control groups (both under stress and irrigated) demonstrate the differential expression of the insert holding the gene p5cs and the aipc promoter, which shows that there was no gene silencing or transgene co-suppression due to a possible presence of multicopies in the different aspects we evaluated. Rooke et al. (2003) observed that polyploidy species, such as wheat, are less prone to gene silencing. This is justified by the large buffering capacity of the inserted genes. The variation we noted between the events, regarding the different physiological parameters observed, can be explained by considering experimental and environmental factors that are not controlled and are not directly associated with the transgene (James et al., 2004; Saint Pierre et al., 2012). After rehydration (Day 20), proline levels decreased, which proved once again the important action of osmolyte during the water stress process (Figure 1B).
Figure 1C shows values obtained for the parameter MSI, under water stress conditions. Under normal conditions of water supply, control and transgenic plants displayed similar results (83-87%). We observed cell membrane injury due to water stress from the 12th and 16th days on after applying water restriction. The lowest values for MSI were found in plants in the control group under stress (40%) when compared to transgenic events: 66% (event 591), 83% (event 14), 78% (event 326), 76% (event 164), and 78% (event 312). After irrigation was carried out again, in the assessment of the 20th day, only the events 591, 326, and 164 presented an MSI close to the irrigated control value (83%). This suggests the maintenance of the integrity of cell membranes. Plants in the control group under water stress and the events 14 and 312 had their MSI near 68%.
MDA content estimated for control and transgenic plants in the different events was similar before water stress started (Figure 1D). After the 12th day, transgenic plants and the stressed control group displayed decreased values of MDA, which were also different from the irrigated control group. The assessment of the 20th day, after rehydration, showed that MDA values had increased again, which can be justified by metabolism acceleration and the onset of senescence, since only plants in the event 591 presented lower and significant MDA levels when compared to the control group. That increase might have happened due to a lack of proline, as it might have moved to reproductive tissues. Therefore, plant cells (control and transgenic) would suffer more damage to oxidative stress because of water deficit (Ozden et al., 2009).
Low MDA content represents low lipid peroxidation. It is, therefore, inversely proportional to the membrane stability (Bhatnagar-Mathur et al., 2009). Data obtained regarding lipid peroxidation, estimated through MDA content, allowed us to conclude that transgenic plants have a better ability to preserve the plant cell intact for a longer period. We were able to prove it on the 20th day of assessment, possibly due to the high levels of proline (observed on the 16th day of evaluation) (Figures 1B and 1D). Several studies have showed that high proline levels in cells might prevent oxidative stress caused by the presence of reactive oxygen species (ROS), which is a result of water stress (Hong-Bo et al., 2006; Hoque et al., 2007; Ahmad et al., 2008; Bhatnagar-Mathur et al., 2009). This condition resulted in a higher maintenance of cellular integrity in the events we evaluated compared to the control group.
Similar to our experiment, Pavei et al. (2016) evaluated 16 different transgenic plants (T2) transformed to p5cs using constitutive promoter (Ubi) and stress induced promoter (aipc) subjected to 12 days of water stress. As results, transgenic plants independently of the promoter used produced approximately 1.85 times more proline than non-transgenic plants, but MDA values did not differ between transgenic plants and control. However, transgenic plants showed a better RWC at the end of evaluation period (84%- transgenic and 57%-control). We can conclude that proline is essential to copy the oxidative stress damage, perhaps the MDA measurement was not sufficient to quantify the antioxidant effect of proline on cells.
Productive indexes indicate the decrease of production in water deficit conditions (Table 1). The irrigated control group presented: a mean of 3 cobs per plant; 138 seeds per plant; 475 mg of weight per 100 seeds; and 3.3 g of seeds total weight per plant. The stressed control group showed a decrease of 33% in the number of cobs, 75% in the mean number of seeds per plant, 34% in the weight of 100 seeds (310.46 mg), and 5.5 times the total weight of seeds (0.6 g). Transgenic plants in the event 14 showed the best productive indexes compared to the stressed control group. They had an increase of 106% in the number of seeds, 1.3 times in the number of cobs, 26% in the weight of 100 seeds, and 116% in the total weight of seeds. Event 312 showed an increase of 1.5 times in the number of seeds, 51% in the number of cobs, 50% in the weight of 100 seeds, and 100% in the total weight of seeds.
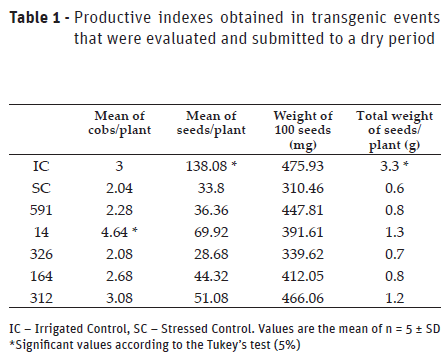
Lemos et al. (2008) observed that the dry period applied in the booting stage of CD200126 plants resulted in a decreased weight of 51.8% of in 100 seeds and of 80% in COB sprouting. After that, severe water stress was applied for 8 days. Saint Pierre et al. (2012), on the other hand, stated that the decrease in plant productivity might be because plants submitted to water stress display a sharp delay in growth. When evaluating transgenic wheat’s (DREB1A) productive performance, they observed that transgenic plants displayed a more conservative profile for plant growth when exposed to reduced water consumption but showed higher survival rates when compared to plants in the control group, with no delay in the culture phenological cycle.
Finally, the results obtained in our study show that proline would act as an osmotic agent, maintaining the membrane integrity, which might be helpful to plants’ survival. However, productivity is considered a polygenic trait in which different genes and environmental factors are involved. Obtaining drought-resistant wheat elite genotypes has been a challenge, as it is a multigenic trait highly affected by environmental conditions (Saad et al., 2013).
Due to the fact that timeline for commercialization of an transgenic event is long (∼14 years for the first commercial launch) (Fraley, 2015) and the investments are quite high (Rudelsheim et al., 2018), biotech trait developers aim to create and identify events that optimally meets the biotech trait product goal in terms of expression, stability, safety, and utility to support commercial release (Mumm, 2013).
CONCLUSIONS
We noticed an increase in proline levels in transgenic plants from the 12th day on, and the highest index was observed in the event 312 and in all transgenic events on the16th day, resulting in higher stability of transgenic plants’ membranes compared to the stressed control group. These results show the osmoprotectant effect of proline on cell membranes. We also observed differential phenotypical characteristics in events containing the gene p5cs and the aipc promoter in the T3 generation, in different events of the 16th day. However, higher levels of proline did not result in higher productivity indexes in transgenic plants.
REFERENCES
Ahmad, P.; John, R.; Sarwat, M. and Umar, S. (2008) - Responses of proline, lipid peroxidation and antioxidative enzymes in two varieties of Pisum sativum L. under salt stress. International Journal of Plant Production, vol. 2, p. 1735-8043. [ Links ]
Anand, A.; Trick, H.N.; Gill, B.S. and Muthukrishnan, S. (2003) - Stable transgene expression and random silencing in wheat. Plant Biotechnology Journal, vol. 1, n. 4, p. 241-251. https://doi.org/10.1046/j.1467-7652.2003.00023.x [ Links ]
Ashraf, M. and McNeill, Y.T. (2008) - Variability in Salt Tolerance of Nine Spring Wheat Cultivars. Journal of Agronomy and Crop Science, vol. 160, n. 1, p. 14–21. https://doi.org/10.1111/j.1439-037X.1988.tb01160.x [ Links ]
Babu, M.; Luscombe, N.M.; Aravind, L.; Gerstein, M. and Teichmann, S.A. (2004) - Structure and evolution of transcriptional regulatory networks. Current Opinion in Structural Biology, vol. 14, n. 3, p. 283-291. https://doi.org/10.1016/j.sbi.2004.05.004 [ Links ]
Bhaskara, G.B.; Yang, T.H. & Verslues, P.E. (2015) - Dynamic proline metabolism: importance and regulation in water limited environments. Frontiers in Plant Science, vol. 6, art. 484. https://doi.org/10.3389/fpls.2015.00484 [ Links ]
Basri, Z. (2005)-Transgene inheritance in transgenic wheat. Jurnal Matematika dan Sains, vol. 10, n. 1, p. 1-9. [ Links ]
Bates, L.S.; Waldran, R.P. and Teare, I.D. (1973) - Rapid determination of free proline for water studies. Plant and Soil, vol. 39, p. 205-208. https://doi.org/10.1007/BF00018060 [ Links ]
Bhatnagar-Mathur, P.; Devi, M.J.; Vadez, V. and Sharma, K.K. (2009) - Differential antioxidative responses in transgenic peanut bear no relationship to their superior transpiration efficiency under drought stress. Journal of Plant Physiology, vol. 166, n. 11, p. 1207-1217. https://doi.org/10.1016/j.jplph.2009.01.001 [ Links ]
Bohorova, N.; Fennell, S.; McLean, S.; Pellegrineschi, A. and Hoisington, D. (1999) - Laboratory protocols. CIMMYT Applied Genetic Engineering Laboratory. Mexico D.F.: CIMMYT, 139p. [ Links ]
Cruz, C.D. (2006) - Programa Genes-estatística experimental e matrizes. 1ed. Viçosa-MG, Editora UFV, vol. 1, 285 p. [ Links ]
Dietz-Pfeilstetter, A. (2010) - Stability of transgene expression as a challenge for genetic engineering. Plant Science, vol. 179, n. 3, p. 164-167. https://doi.org/10.1016/j.plantsci.2010.04.015 [ Links ]
Dolferus, R.; Ji, X.M. and Richards, R.A. (2011) -Abiotic stress and control of grain number in cereals. Plant Science, vol. 181, n. 4, p. 331-341. http://dx.doi.org/10.1016/j.plantsci.2011.05.015 [ Links ]
Dunwell, J.M. (2014) - Transgenic cereals: Current status and future prospects. Journal of Cereal Science, vol. 59, n. 3, p. 1-16. http://dx.doi.org/10.1016/j.jcs.2013.08.008 [ Links ]
Fraley, R. (2015) - Presentation to the National Academy of Sciences. <https://vimeo.com/album/3192610/video/115717420> [ Links ]
Hong-Bo, S.; Xiao-Yan, C.; Li-Ye, C.; Xi-Ning, Z.; Gang, W.; Yong-Bing, Y.; Chang-Xing, Z. and Zan-Min, Z. (2006) - Investigation on the relationship of proline with wheat anti-drought under soil water deficits. Colloids and Surfaces B: Biointerfaces, vol. 53, n. 1, p. 113-119. https://doi.org/10.1016/j.colsurfb.2006.08.008 [ Links ]
Hoque, M.A.; Okuma, E.; Banu, M.N.A.; Nakamura, Y.; Shimoishi, Y. and Murata, Y. (2007) - Exogenous proline mitigates the detrimental effects of salt stress more than exogenous betaine by increasing antioxidant enzyme activities. Journal of Plant Physiology, vol. 164, n. 5, p. 553-561. https://doi.org/10.1016/j.jplph.2006.03.010 [ Links ]
James, V.A.; Worland, B.; Snape, J.W. and Vain, P. (2004) - Strategies for precise quantification of transgene expression levels over several generations in rice. Journal of Experimental Botany, vol. 55, n. 401, p. 1307-1313. https://doi.org/10.1093/jxb/erh133 [ Links ]
Khan, M.S.; Ahmad, A. & Khan, M.A. (2015) - Utilization of genes encoding osmoprotectants in transgenic plants for enhanced abiotic stress tolerance. Electronic Journal of Biotechnology, vol. 18, n. 4, p. 257–266. https://doi.org/10.1016/j.ejbt.2015.04.002 [ Links ]
Lemos, J.M.; Vendruscolo, E.C.G.; Schuster, I. and Santos, M.F. (2011) - Physiological and biochemical responses of wheat subjected to water deficit stress at different phonological stages of development. Journal of Agricultural Science and Technology B, vol. 1, n. 8, p. 1116-1124. [ Links ]
Meng, L.; Ziv, M. and Lemaux, P.G. (2006) - Nature of stress and transgene locus influences transgene expression stability in barley. Plant Molecular Biology, vol. 62, n. 1-2, p. 15-28. https://doi.org/10.1007/s11103-006-9000-7 [ Links ]
Mlynarova, L.; Keizer, L.C.P.; Stiekema, W.J. and Nap, J.P. (1996) - Approaching the low limits of transgene variability. The Plant Cell, vol. 14, p. 129-135. http://dx.doi.org/10.1105/tpc.8.9.1589 [ Links ]
Mumm, R.H. (2013) - A Look at Product Development with Genetically Modified Crops: Examples from Maize. Journal of Agricultural and Food Chemistry, vol. 61, n. 35, p. 8254−8259. https://doi.org/10.1021/jf400685y [ Links ]
Munns, R. and Tester, M. (2008) - Mechanisms of Salinity Tolerance. Annual Review of Plant Biology, vol. 59, p. 651-681. https://doi.org/10.1146/annurev.arplant.59.032607.092911 [ Links ]
Ozden, M.; Demirel, U. and Kahraman, A. (2009) - Effects of proline on antioxidant system in leaves of grapevine (Vitis vinifera L.) exposed to oxidative stress by H2O2. Scientia Horticulture, vol. 119, n. 2, p. 163-168. https://doi.org/10.1016/j.scienta.2008.07.031 [ Links ]
Pavei, D.; Gonçalves-Vidigal, M.C.; Schuelter, A.R.; Schuster, I.; Vieira, E.S.N.; Vendruscolo, E.C.G. & Poletine, J.P. (2016) - Response to water stress in transgenic (p5cs gene) wheat plants (Triticum aestivum L.). Australian Journal of Crop Science, vol. 10, n. 6, p.776-783. https://doi.org/10.21475/ajcs.2016.10.06.p7000 [ Links ]
Rooke, L.; Steele, S.H.; Barcelo, P.; Shewry, P.R. and Lazzeri, P.A. (2003) - Transgene inheritance, segregation and expression in bread wheat. Euphytica, vol. 129, n. 3, p. 301-309. https://doi.org/10.1023/A:1022296017801 [ Links ]
Rüdelsheim, P.; Dumont, P.; Freyssinet, G.; Pertry, I. & Heijde, M. (2018) - Off-Patent Transgenic Events: Challenges and Opportunities for New Actors and Markets in Agriculture. Frontiers in Bioengineering and Biotechnology, vol. 6, art. 71. https://doi.org/10.3389/fbioe.2018.00071 [ Links ]
Saad, A.S.I.; Li, X.; Li, H.P.; Huang, T.; Gao, C.G.; Guo, M.W.; Cheng, W.; Zhao, G.Y. and Liao, Y.C. (2013) - A rice stress-responsive NAC gene enhances tolerance of transgenic wheat to drought and salt stresses. Plant Science, vol. 203-204, p. 33-40. https://doi.org/10.1016/j.plantsci.2012.12.016 [ Links ]
Saint Pierre, C.; Crossa, J.L.; Bonnett, D.; Yamaguchi-Shinozaki, K. and Reynolds, R.P. (2012) - Phenotyping transgenic wheat for drought resistance. Journal of Experimental Botany, vol. 63, n. 5, p. 1799-1808. https://doi.org/10.1093/jxb/err385 [ Links ]
Sairam, R.K.; Desmukh, P.S. and Saxena, D.C. (1998) - Role of antioxidant systems in wheat genotypes tolerant to water stress. Biologia Plantarum, vol. 41, n. 3, p. 387-394. https://doi.org/10.1023/A:1001898310321 [ Links ]
Schonfeld, M.A. (1988) - Water relations in winter wheat as drought resistance indicators. Crop Science, vol. 28, n. 3, p. 526-531. https://doi.org/10.2135/cropsci1988.0011183X002800030021x [ Links ]
Ulker, B.; Allen, G.C.; Thompson, W.F. and Spiker, S. (1999) - A tobacco matrix attachment region reduces the loss of transgene expression in the progeny tobacco plants. The Plant Journal, vol. 18, n. 3, p. 253-263. https://doi.org/10.1046/j.1365-313X.1999.00453.x [ Links ]
Vendruscolo, E.C.G.; Schuster, I.; Pileggi, M.; Scapim, C.A. and Molinari, H.B.C. (2007) - Stress-induced synthesis of proline confers tolerance to water deficit in transgenic wheat. Journal of Plant Physiology, vol. 164, n. 10, p. 1367-1376. http://dx.doi.org/10.1016/j.jplph.2007.05.001 [ Links ]
Xiao, B.Z.; Chen, X.; Xiang, C.B.; Tang, N.; Zhang, Q.F. and Xiong, L.Z. (2009) - Evaluation of seven function-known candidate gens for their effects on improving drought resistance of transgenic rice under field conditions. Molecular Plant, vol. 2, n. 1, p. 469-490. https://doi.org/10.1093/mp/ssn068 [ Links ]
Yao, Q.; Cong, L.; Chang, J.L.; Li, K.X.; Yang, G.X. and He, G.Y. (2006) - Low copy number of gene transfer and stable expression in a commercial wheat cultivar via particle bombardment. Journal of Experimental Botany, vol. 57, n. 14, p. 3737-3746. https://doi.org/10.1093/jxb/erl145 [ Links ]
Zadocks, J.C.; Chang, T.T. and Konzak, C.F. (1974) - A decimal code for growth stages of cereals. Weed Research, vol. 14, n. 16, p. 415-421. https://doi.org/10.1111/j.1365-3180.1974.tb01084.x [ Links ]
Zhu, B.C.; Su, J.; Chan, M.C.; Verma, D.P.S.; Fan, Y.L. and Wu, R. (1998) - Overexpression of Δ1- pyrroline-5-carboxylate synthetase gene and analysis of tolerance to water- and salt-stress in transgenic rice. Plant Science, vol. 139, n. 1, p. 41-48. https://doi.org/10.1016/S0168-9452(98)00175-7 [ Links ]
ACKNOWLEDGEMENTS
We would like to thank Coodetec and Capes for their financial support by granting the first author a scholarship. We would also like to thank the Academic Publishing Advisory Center (Centro de Assessoria de Publicação Acadêmica – CAPA) of the Federal University of Paraná for the assistance with English language editing. The experiment was carried out in the Biotechnology Laboratory and the Plant House of Coodetec that have CQB.
Received/recebido: 2018.09.16
Aceite/accepted: 2019.02.03